
Variations in intracellular large anion content mean that intracellular Cl – concentration varies considerably. Cl – will diffuse down this concentration gradient back into the cell.Īn asymmetric equilibrium is formed with the electrical and concentration gradients opposing each other – this is Donnan’s equilibrium.ĭonnan’s Equilibrium: ‘The presence of a charged impermeant ion (anionic protein) on one side of a semi-permeable membrane will result in an asymmetric distribution of permeant charged ions (Cl –)’. This creates a concentration gradient with higher concentration of Cl – outside of the cell. phosphate) force Cl – out of the cell down an electrical gradient. These, along with other large anions trapped in the cell (e.g. Intracellular proteins are negatively charged at physiological pH. Cl – and the Donnan EquilibriumĬl – ions are differentially distributed across the cell membrane due to their negative charge and because they can move freely across the cell membrane via protein channels. Intracellular Ca 2+is either actively transported out of the cell or into the endoplasmic reticulum/mitochondria – this leaves very low levels in the cell. The Na +-K + ATPase carrier protein has to be phosphorylated by one ATP molecule. It pumps 3 Na + ions out of the cell, and 2 K + into the cell. 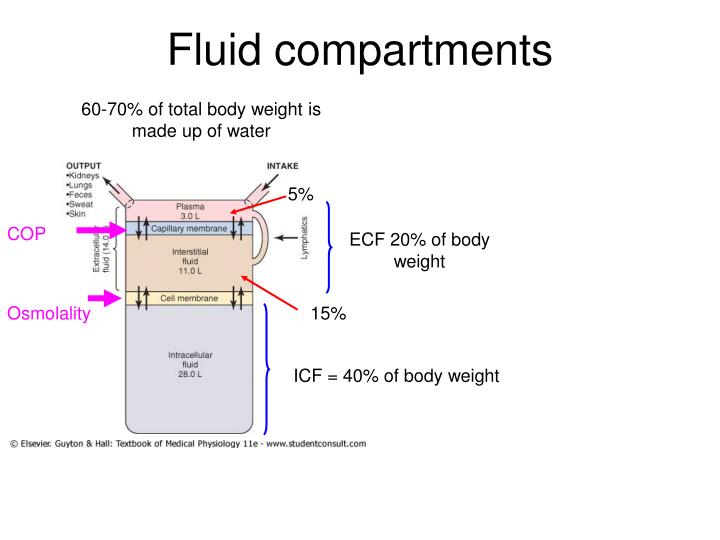
The Na +-K + ATPase pump acts to maintain the cellular environment by actively moving Na + and K + against their concentration gradient. 
This is prevented by the activity of the Na +-K + ATPase pump. Without intervention, this would eventually lead to equalisation of both ions on both sides of the membrane, which would be physiologically non-viable. K + and Na + follow their concentration gradients (K + out of the cell, Na + in). Na + concentration is much higher extracellularly.K + concentration is much higher intracellularly.The most important difference between ICF and ECF is the relative concentration of cations: The biochemical balance between ICF and ECF is very tightly controlled as many crucial physiological processes depend on its composition. It includes CSF, exocrine secretions and gastrointestinal secretions. * Transcellular fluid is the name given to fluids in the body that are not contained within the ICF or ISF/plasma (ECF) compartments. These two compartments are separated by cell membranes, and are very different in their composition (i.e. Extracellular fluid (ECF) – itself can be divided into:.Intracellular fluid (ICF) – the fluid within cells which accounts for 65% of total body weight.Water makes up 50-70% of body mass – approximately 42L in a 70kg person. The osmolality of plasma = 290 mosmol/kg H 2O Body Water Compartments Osmolality is the preferred expression of physiologists because it is independent of temperature and pressure. Osmolality – osmotic concentration per volume of solution (osmol/kg H 2O).Osmolarity – osmotic concentration per mass of solvent (osmol/L).It can be expressed as ‘osmolarity’ or ‘osmolality’: Osmotic potential is determined by the number of osmotically active particles per litre. Fluid at lower osmotic potential is hypotonic.Fluid at higher osmotic potential is hypertonic. 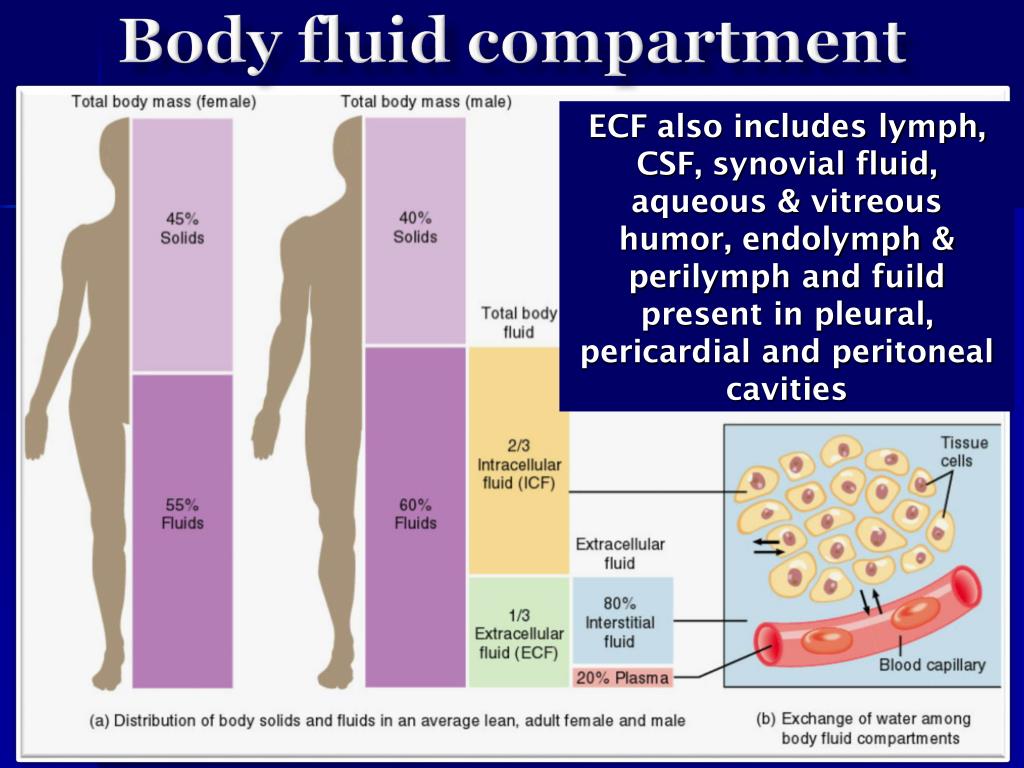
Fluid at the same osmotic potential as plasma is hypotonic.The creation of osmotic gradients is the primary method for the movement of water in the body. ‘Osmosis is the movement of water across a semi-permeable membrane from regions of low solute concentration to those of higher concentration’.īiological membranes are semi-permeable – they allow the free movement of water but restrict the movement of solutes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
